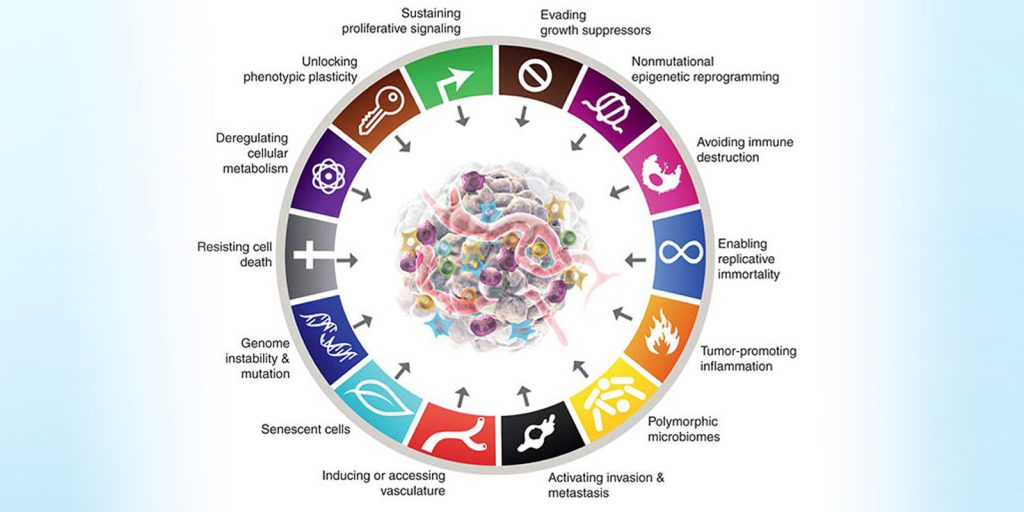
Over the past few decades, our understanding of cancer has shifted from viewing it as a single disease to recognizing it as a complex set of diseases driven by specific biological capabilities. In their seminal papers published in 2000, 2011, and updated again in 2022, Douglas Hanahan and Robert Weinberg proposed a conceptual framework known as the “Hallmarks of Cancer”, which describes the essential biological traits that cancer cells acquire during tumor development.
These hallmarks include properties such as sustaining proliferative signaling, evading growth suppressors, resisting cell death, enabling replicative immortality, inducing angiogenesis, and activating invasion and metastasis. In 2011, additional hallmarks and enabling characteristics were added, such as reprogramming of energy metabolism and avoiding immune destruction. In the most recent 2022 update, Hanahan introduced further refinements, including phenotypic plasticity, nonmutational epigenetic reprogramming, polymorphic microbiomes, and influences from senescent cells. These hallmarks provide an organizing principle to understand how normal cells transform into malignant ones.
The Hallmarks of Cancer as conceptualized by Hanahan & Weinberg
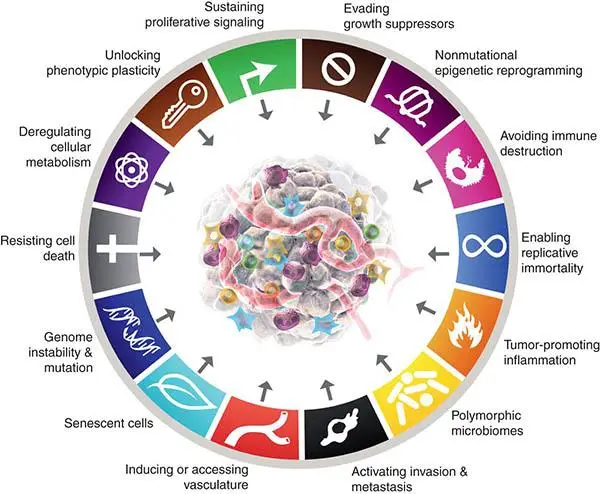
Understanding these hallmarks has fundamentally shaped modern oncology by revealing the underlying mechanisms that drive cancer progression—and more importantly, it has paved the way for precision medicine. Instead of using traditional therapies that affect both healthy and cancerous cells, we now aim to design therapies that specifically target the aberrant pathways and immune escape mechanisms used by cancer cells.
This is where targeted therapy and immunotherapy come into play.
Targeted therapies are designed to interfere with specific molecules or signaling pathways that are crucial for cancer cell survival and proliferation
Linking Hallmarks to Therapies
| Hallmark / Enabling Trait | Therapeutic Strategy | Examples |
|---|---|---|
| Sustaining proliferative signaling | Target growth-factor pathways | EGFR inhibitors, HER2 antibodies |
| Evading growth suppressors | Inhibit cell-cycle regulators | CDK4/6 inhibitors |
| Resisting cell death | Promote apoptosis | BCL-2 inhibitors |
| Inducing/accessing vasculature | Block angiogenesis | VEGF inhibitors |
| Deregulating metabolism | Disrupt tumor metabolism | Glycolysis inhibitors; metabolic modulators |
| Enabling replicative immortality | Target telomerase pathways (emerging) | Under clinical investigation |
| Avoiding immune destruction | Unleash T-cell response | Checkpoint inhibitors (PD-1, PD-L1, CTLA-4) |
| Unlocking phenotypic plasticity / Epigenetic reprogramming | Sensitize tumors to immune or targeted therapies | Emerging combination strategies |
| Genome instability / inflammation | Exploit DNA damage pathways | PARP inhibitors in BRCA-mutant cancers |
Immunotherapies harness the patient’s own immune system to recognize and destroy cancer cells
5 Main Types of Cancer Immunotherapy
| Type | Description | Examples |
|---|---|---|
| Immune Checkpoint Inhibitors | Block proteins (e.g., PD-1, CTLA-4) that restrain T-cells | Pembrolizumab (PD-1), Ipilimumab (CTLA-4) |
| CAR T-cell Therapy | Patient’s T cells are genetically modified to attack tumor cells | Tisagenlecleucel (CD19), Axicabtagene ciloleucel |
| Cancer Vaccines | Stimulate the immune system to target tumor-specific antigens | Sipuleucel-T for prostate cancer |
| Monoclonal Antibodies | Target tumor antigens or flag cells for destruction | Rituximab (CD20), Trastuzumab (HER2) |
| Cytokine Therapy | Use immune-stimulating proteins to boost immune response | IL-2, Interferon-α |
Together, these therapies represent a paradigm shift—from a “one-size-fits-all” approach to personalized, mechanism-based treatments guided by the molecular and immunological hallmarks of each individual’s cancer.
References
- Hanahan D, Weinberg RA. Hallmarks of Cancer. Cell. 2000;100(1):57–70.
- Hanahan D, Weinberg RA. Hallmarks of Cancer: The Next Generation. Cell. 2011;144(5):646–74.
- Hanahan D. Hallmarks of Cancer: New Dimensions. Cancer Discovery. 2022;12(1):31–46.










